Not sure what to study, or if you are going to pass? We can help!
I'm a teacher candidate at a university/college I'd like to transition to teaching I'm a current / former teacher I'm in leadership for K-12 or Higher Ed (EPP)Overview
Practice Test Links
TExES Science 7-12 Ultimate Guide and Practice Test
Preparing to take the TExES Science 7-12?
Awesome!
You’ve found the right page. We will answer every question you have and tell you exactly what you need to study to pass the TExES Science 7-12.
TExES Science 7-12
-
Quick Facts
-
Domain I: Scientific Inquiry and Processes
-
Domain II: Physics
-
Domain III: Chemistry
-
Domain IV: Cell Structure and Processes
-
Domain V: Heredity and Evolution of Life
-
Domain VI: Diversity of Life
-
Domain VII: Interdependence of Life and Environmental Systems
-
Domain VIII: Earth’s History and the Structure and Function of Earth Systems
-
Domain IX: Components and Properties of the Solar System and the Universe
-
Domain X: Science Learning, Instruction, and Assessment
-
Practice Questions and Answers
TExES Science 7-12 Quick Facts
This examination determines if the test taker has the knowledge to be an entry-level science teacher in Texas’ public school system. The test over Science 7-12 encompasses ten domains.
Cost:
The exam costs $116.
Scoring:
Exams are scored on a scale of 100 to 300. A passing score for certification is 240.
There are “pilot” questions on the test which are experimental and only designed to determine whether or not they are good questions for future tests. There is no way of knowing which questions are pilot questions and which are part of your test. This is why it is imperative to answer every question to the best of your ability. If you are unsure, eliminate the answer choices you know are wrong and make your best guess from the remaining answers.
Study time:
In order to pass, you should break the test topics down into your strengths and weaknesses. Give yourself enough time to work on each topic until you feel confident with the majority of the subjects. Use 240Tutoring’s TExES Science 7-12 study guide and create a study plan detailing the time you need to study each topic and the days you plan to do it.
What test takers wish they would’ve known:
-
A Periodic Table is provided for your reference. Do not spend time memorizing numbers on the Periodic Table, just understand how to interpret data presented to you on the table. Know what the numbers represent and understand the classification of groups and families as they pertain to rows and columns in the table.
-
There is more than one version of this test. Each one tests the same overall content with the same number of questions allotted for each topic, but they do vary in question subject matter. For example, you might be asked to create a hypothesis based on an experiment about plants while someone else is asked to create a hypothesis on an experiment about pharmaceuticals. The subject matter varies, but both these questions are testing the same concept: how well do you understand the scientific method?
-
As with any timed exam, pace yourself. Your score is calculated based on what you answer correctly, so leaving any question blank only hurts that score. If you don’t know the correct answer with certainty, eliminate the answers you know to be incorrect, then select what you believe to be the best answer.
Information and screenshots obtained from the National Evaluation Series’
website
.
Overview
This domain has about 14 multiple-choice questions. These questions account for 10% of the entire exam.
So, let’s talk about a few of the concepts that you will likely see within this
domain.
Standard Safety Equipment
A typical laboratory is equipped with both (A) equipment to navigate an accident in the event that it happens, as well as (B) personal protective equipment (PPE) to proactively keep staff and students protected from materials they encounter on a day-to-day basis while doing lab work.
Some of the standard equipment you will see is a fire extinguisher and fire blanket. In the event that something does catch on fire, an extinguisher can control small flames until public safety professionals can arrive on the scene to assist. The fire blanket is made of a non-flammable material that can be used to put out flames on a person because a fire extinguisher is not ideal for use on a person
A lab setting should include an eyewash station or at least a faucet-mounted eyewash. In the event that someone gets any chemical in their eyes, this is a simple and very effective way to safely wash it out with water. An individual simply leans over the eyewash device and turns on the water pressure. The eyes are flushed with water for a period of 15 minutes. Often times in lab settings, the room is also equipped with a safety shower which is used if any corrosive or toxic chemical is spilled on clothing. In that event, an individual would stand under the safety shower and rinse their clothing for a period of 10-15 minutes until they can remove the clothing.
Personal protective equipment should be stocked in any lab setting to adequately protect the skin and clothing of the maximum number of individuals in the classroom at any one time. This would include disposable gloves (latex and nitrile), lab safety goggles, and lab coats or lab aprons. If there are any methods by which you heat anything in the lab, at least one pair of heavy duty, heat resistant gloves should also be kept in the lab for handling hot or potentially hot objects, as disposable gloves will not suffice for protection from high temperatures.
It should be noted in the classroom setting and somewhere in the room that close-toed shoes and fully clothed legs are required in a lab setting. Long hair should always be tied securely back. It is highly recommended that teachers have students sign off on these safety measures at the beginning of the course to verbalize understanding of these lab safety precautions and to demonstrate their agreement with compliance.
Types of Scientific Investigations
In a descriptive investigation
, the purpose is to draw conclusions about something or describe it. Since it is essentially a learning process about something, it is typically used when the researchers know little about the subject matter. It poses a question to be answered but does not typically include a hypothesis. Keywords to look for when identifying a descriptive investigation include
describe, identify, observe,
and
list
.
An example of a descriptive investigation would be students learning basic information about rock formation. The student may ask, “how do plutonic rocks form beneath the Earth’s surface?” The experiment process would then include research of the topic, data collection from this research, and some type of conclusion drawn from the investigative process. The student would learn that molten rock from the Earth’s mantle can erupt from a volcano and force the hot, liquid magma onto the earth’s surface where it cools and becomes solid volcanic rock. With little prior knowledge of the subject matter, the student isn’t likely to pose a hypothesis in a descriptive study, but can ultimately answer a question utilizing this type of investigation.
In a comparative investigation
, the purpose is to compare/contrast the relationship between two things. Keywords to look for when identifying a comparative relationship include
categorize, compare and contrast,
or
differentiate between
.
While a comparative investigation typically includes components of the scientific inquiry such as observations and a scientific question, it also includes a hypothesis, unlike the descriptive investigation. Since it compares two systems, or two populations, or two conditions to one another, the researcher generally has some basic knowledge that leads them to believe one has an edge over the other. This knowledge is what prompts the experiment.
For instance, one spring day, you park your car under a tree mid-morning. When you go to leave for lunch, you notice your car is a bit chilly. The next day, you park your car in an open area, exposed to sunlight. When you leave for lunch, you feel the car is warm. This observation leads you to compare the difference in temperatures at the same time between point A (under the tree) and point B (an open area, uninhabited by shade trees). Another example is you notice the pH of the water in your family pond changes following a heavy rain. You might consider the effects of rain on the pH level by measuring it before and after the rain.
The difference between a comparative relationship investigation and an experimental investigation is the presence of a control group. Since a control group is defined as a group within the experiment that doesn’t receive treatment by the researchers, we can see that this does not apply to the experiments mentioned above, nor is a control group necessary in either scenario to accurately address the posed questions.
Finally, in experimental investigations
, the same components of the descriptive and comparative investigations are included: a purpose or posed question, a hypothesis, and independent/dependent variables. But this type of investigation also includes a control group. This type of investigation revolves around the design of an unbiased and fair experiment in which variables are manipulated and controlled to gather evidence which either supports or rejects your hypothesis.
For instance, someone plants a strawberry plant that never produces any strawberries. The following year, they plant it in a different location that receives more sunlight and it produces some strawberries. They then form the hypothesis that “a strawberry plant will produce more fruit when routinely exposed to more hours of sunlight.” Ultimately, this individual is studying the relationship between the sun and a strawberry plant and investigating if sunlight exposure plays a role in fruit production.
These types of experiments have a definite independent and dependent variable. So, for the strawberry plant example, the plant itself is your dependent variable. The sun is the independent variable. The plant may or may not be dependent on the sunlight. To incorporate the control within this experiment, you can place the strawberry plant in an area where it receives an average amount of sunlight per day. So, your control may be a strawberry plant exposed to an average of 4-6 hours of sunlight daily. The variable of sunlight exposure is then controlled by having a strawberry plant sitting on a dark porch, unexposed to any sunlight. The third strawberry plant is exposed to 6-8+ hours of sunlight daily.
In an experimental investigation, every other identified variable must be held constant to deduce that the sunlight is the only variable manipulating the outcome of the experiment. So, it would be imperative to make sure all three strawberry plants were being watered at the same times, with the same amount of water. If fertilizer is utilized, all 3 plants should receive the same amount of fertilizer, at the same frequency and at the same time. All 3 strawberry plants must be the same brand, or grown in the same setting, and planted in the same type of soil. To accurately produce an experimental investigation, the researcher must identify all variables and control all of them but one, in this case, sunlight.
Overview
This domain has about 28 multiple-choice questions. These questions account for 20% of the entire exam.
Let’s discuss a few physics concepts.
Newton’s Laws of Motion
Newton’s First Law
: Every object remains at rest or in uniform motion in a straight line unless compelled to change its state of motion by the action of an external force.
What does that mean? Essentially, any object will move in a straight line, unless something interferes with that trajectory. That “something” can be a hill, friction, some type of barrier, etc. For instance, if I roll a marble across a wooden floor, it will keep rolling, unless something opposes that motion. If it rolls until it reaches a staircase, it will accelerate more quickly when it drops off a stair. That stair was an external force acting upon the marble. If the marble is rolling across a wooden floor and encounters a rug, there will be an introduction of friction, which becomes a force affecting the action of the marble. If the marble rolls across the floor and then encounters a piece of furniture, or some type of barrier, that straight line of motion was interfered with.
Newton’s Second Law
: Force is equal to the change in momentum per change in time. For a constant mass, force equals mass times acceleration (F = ma).
What does that mean? This law addresses the role of mass relative to force. Force is inversely proportional to mass. If one number goes up, the other must come down and vice versa. This makes sense if you consider the following scenario. If I place two balls at the top of the same, huge hill and push them both at the same time, you’d expect that they’d behave in the same fashion (assuming the balls were identical).
But let’s assume one of those is a bowling ball and the other is a plastic, bouncy ball. The force at which both of those balls have while descending the hill varies greatly. This is simply due to the difference in their masses. The acceleration of both these balls also varies greatly as a direct result of their varying masses. The plastic, bouncy ball will accelerate with less force and require less force to stop it. The bowling ball will descend the hill with greater force, because of its dense mass. Don’t believe me? Try it. And get out of the way when that bowling ball comes down the hill! Essentially, Newton’s second law addresses the role that mass plays in affecting force and acceleration.
Newton’s Third Law
: For every action, there is an equal and opposite reaction.
What does that mean? Essentially, it means that force occurs in pairs. For instance, you sit down at your desk in a chair. As you sit in the chair, a force is applied to the chair pushing it downward. If that were the only force occuring at that time, the chair would collapse, but because the chair applies an equal and opposite force upward, it remains intact.
Of course, the response of the equal and opposite force varies depending on the mass of the force, as we learned from the second law. So, if an 18-wheeler truck crashes into a compact car, the compact car in turn also exerts a force back onto the 18-wheeler. Due to the greater mass of the 18-wheeler, the force involved in the “equal and opposite” reaction will vary.
Entropy
Entropy measures the amount of disorder in a system. The second law of thermodynamics tells us that total entropy of a closed system can never decrease over time. So essentially, the entropy of the universe can only increase. Why? Because the universe is the supreme, closed system since there is nothing outside of it for it to react with thermodynamically.
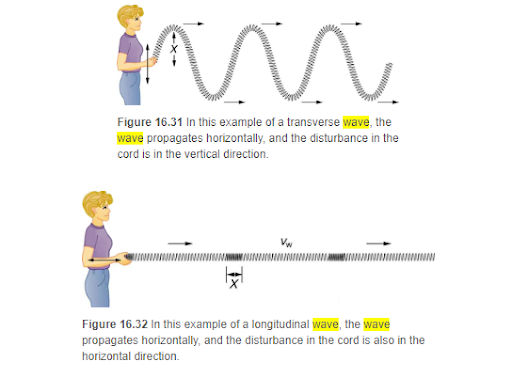
Transverse and Longitudinal Waves
The difference in longitudinal and transverse waves lies in the difference in the direction of propagation of the wave relative to the direction the particles of the medium are moving. Whew! Don’t over think this one! As the name implies, in a longitudinal wave, the waves move in the same parallel direction as the particles of the medium are moving. Both are going in a straight, parallel trajectory. Conversely, in a transverse wave, the particles of the medium move perpendicular to the direction the wave is moving.
Overview
This domain has about 28 multiple-choice questions. These questions account for 20% of the entire exam.
Take a look at these chemistry concepts that are likely to be on the test.
Charles’s Law
Charles’s Law states that the volume of an ideal gas at a constant pressure is directly proportional to the absolute temperature. As a reminder, pressure exists when gas molecules move about rapidly. The more rapidly they move, the higher the resulting pressure. The smaller a container, the more rapidly the gas molecules are moving. The larger the volume of the container, the more room these gas molecules have to move about freely, which decreases pressure. So, if the temperature of a gas increases, the pressure will remain unchanged only if the volume of the container in which the gas is contained increases also. This happens as a result of the gas molecules being required to travel greater distances when the volume of the container increases. The surface area of the container also increases when the volume does, meaning there is more room for the travel of the gas molecules when the volume of the container increases, keeping the pressure from increasing. The higher temperature yields greater kinetic energy which in turn balances out the number of molecule-wall collisions and molecule-molecule collisions.
Stable and Unstable Isotopes
An isotope is an element that has the same number of protons, and thus the same atomic number, but varies in the number of neutrons, and thus has a different atomic weight or atomic mass.
Generally speaking, the larger the difference in the element’s number of neutrons relative to its number of protons, the more unstable the element becomes. This is because it becomes increasingly likely that it will shed its “extra” neutrons to achieve more stability.
For example, the isotope carbon occurs as carbon-12, carbon-13, and carbon-14. In all 3 elements, there are 6 protons. Since the atomic weight of carbon is approximately 12, we know that carbon, in its typical state, also has 6 (atomic weight (12) – atomic number (6) = 6) neutrons. When carbon exists as carbon-13, there is one extra neutron, or 7 neutrons, and when it occurs as carbon-14, there are 8 neutrons. The increasing number of neutrons results in an unstable atomic nucleus and is thus radioactive.
Acids, Bases, and Salts
Acids are electrolytes that release hydrogen ions (
H
+
). Bases are electrolytes that release ions that combine with hydrogen ions (
H
+
). Bases tend to react with acids to achieve a more neutral state. This results in the formation of water and salts. A salt is an electrolyte that forms when an acid and a base react with one another.
The hydrogen ion concentration is measured by a pH scale. On a pH scale, which ranges from 0-14, each number represents a ten-fold difference in the concentration of hydrogen ions. As the hydrogen ion concentration increases, the pH value decreases.
Overview
This domain has about 11 multiple-choice questions. These questions account for 8% of the entire exam.
The following two concepts are very likely to pop up on the test.
Basic Components of Prokaryotic and Eukaryotic Cells
Prokaryotic cells are single-celled organisms that do not possess a nucleus and do not contain membrane-bound organelles. Prokaryotic cells possess four parts: a plasma membrane, cytoplasm, DNA, and ribosomes. Eukaryotic cells do possess a nucleus and do have membrane-bound organelles (mitochondria, endoplasmic reticulum, Golgi complex, etc.). The DNA found within prokaryotes is circular and prokaryotes reproduce via binary fission, utilizing a single chromosome. Eukaryotes contain linear DNA and reproduce via meiosis or mitosis utilizing a pair (diploid) of rod-shaped chromosomes. Finally, the two differ in size. Prokaryotic cells are significantly smaller (0.1 – 5.0 µm ) than eukaryotic cells which range in size from 10-100 µM.
In conclusion, eukaryotic cells are more complex than the more simple prokaryotic cells.
Anaerobic and Aerobic Respiration
Cellular respiration is the process by which cells produce adenosine triphosphate (ATP), which is the energy currency necessary for them to carry out the processes they utilize to survive. Cellular respiration occurs either anaerobically or aerobically. Aerobic respiration requires oxygen to occur while anaerobic processes do not require oxygen. Aerobic processes occur in the mitochondria of cells since they require oxygen while anaerobic processes occur in the cytosol of cells. While both processes have the same end result of producing ATP, aerobic respiration does so more efficiently in the presence of oxygen.
Overview
This domain has about 11 multiple-choice questions. These questions account for 8% of the entire exam.
Check out the following concepts.
Meiosis and Mitosis
Mitosis is a process by which cells reproduce. Through a series of steps, mitosis produces two genetically-identical daughter cells. Prior to mitosis, during interphase, the cell is considered to be in “neutral”, or its normal state, and is not reproducing. Chromosomes appear as visible chromatin fibers and centrioles are present. As mitosis begins in prophase, the chromosome condenses and centrioles have replicated and moved to opposite poles of the cell. The nucleolus and nuclear envelope disappear while spindle fibers pair with chromosomes and centrioles. Next is metaphase, where the chromosomes line up between centrioles. In anaphase, centromeres begin to pull apart by their spindle fibers and chromosomes migrate in opposite directions. Finally, in telophase, the chromosomes become chromatin, and a nuclear envelope redevelops. In mitosis, nuclear components are divided into two in karyokinesis and then cytoplasm splits in two, which is cytokinesis. Cytokinesis begins in anaphase and continues throughout telophase.
Meiosis, another type of cellular reproduction, yields only one cell and ultimately reduces the number of chromosomes by half. Meiosis is the precursor for human sexual reproduction and undergoes two successive divisions. Starting out, each diploid cell contains two sets of chromosomes, that is, 23 homologous chromosome pairs. These are separated from one another in meiosis I. Following meiosis I, each cell will contain one chromosome from each homologous pair, making them haploid, or containing one set of chromosomes. In meiosis II, chromatids separate and each chromatid is now independent. During prophase of meiosis I, chromosomes shorten and thicken, the nucleoli disappear, and the membrane dissociates. Microtubules assemble the spindle which will be used to separate the chromosomes later. The chromosomes pair up and tightly coil. As metaphase I begins, chromosomes line up between the poles of the newly developing spindle, and each chromosome will ultimately attach to a spindle.
Next is anaphase I where the homologous chromosomes separate and replicated members migrate to respective spindles. Finally in telophase I, the cell divides into two and nuclear membrane reforms around the chromosomes. Meiosis II follows telophase I. In prophase II, chromosomes reappear, replicated. They migrate to positions between the poles of the newly formed spindle. In metaphase II, chromosomes that were replicated attach to spindle fibers and in anaphase II, the centromeres pull apart allowing the chromatids to migrate to opposite poles and the chromatids are now chromosomes. Finally, in telophase II, the two cells formed in meiosis I divide to form two more cells.
While meiosis undergoes similar stages to mitosis, the end result is different.
In summary, Meiosis produces four genetically identical cells, each of which contains half the chromosomes relative to the parent. Mitosis produces only two genetically identical cells, each containing the same number of chromosomes as their parent.
Processes That Contribute to Speciation
Natural Selection is the innate ability of organisms to adapt to their surroundings and their environment in order to survive and reproduce. Generally speaking, organisms that more readily adapt to their surroundings have a better chance of surviving than those that are less adaptable. Furthermore, these organisms that better adapt to their surroundings produce offspring that are more likely to survive.
For example, a population of beetle moves into a new area. The beetle population is about 50% green and 50% tan. Against the foliage background, the tan beetles stand out more so than the green beetles. These tan beetles begin to become a regular snack for predatory animals in the area. As a result, the population of beetles becomes increasingly green, rather than tan. Natural selection, in this case, will likely result in future generations of this beetle becoming increasingly green in color and perhaps the eventual extinction of that tan beetle in that area. These same tan beetles would better survive in a tan rock or cliff setting, where their green counterparts might be more visible to other predatory animals.
Reproductive isolation is the ability of organisms to interbreed. There can be physical barriers between species that prevent breeding, like a river or a mountain range. Or there can be differences in mating rituals or mating seasons which result in reproductive isolation and contribute to speciation.
The Founder Effect occurs when a reduction in genetic diversity happens following some event and the result is a less genetically varied population. Despite the diversity of the original population, the new population will only resemble the founders of a new, smaller population.
All of these things contribute to speciation, which is the formation of distinct, new species. All of the aforementioned processes force species to adapt and evolve in order to survive. The resulting populations which do survive the changes, give rise to new lineages and evolved, more adapted species.